Machine learning accelerates discovery of new 2D structure of titanium dioxide
by Carey Sargent, EPFL, NCCR MARVEL
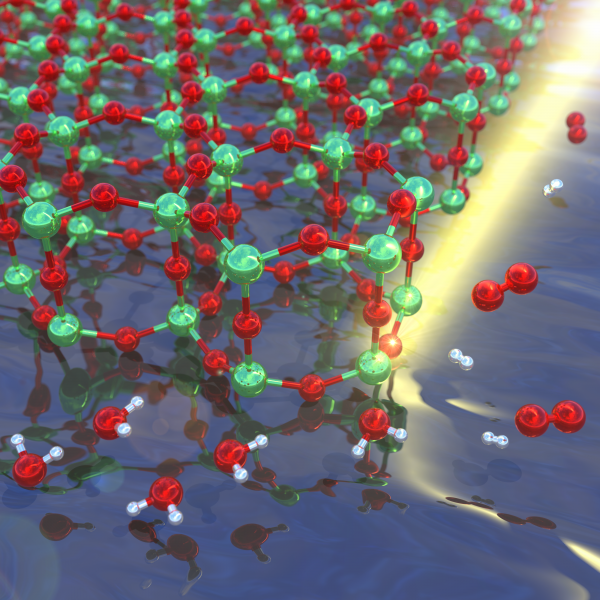
The image shows the two-dimensional TiO2 structure with its hexagonal lattice in the top left with the incident photons represented by a beam of light. As water molecules enter from the bottom left, they dissociate into their constituents, O2 and H2.
Credits: Maximilian Amsler. A similar image was published as the cover of Chemistry of Materials.
“We’re now in a new era where we can make predictions and virtually discover new materials on the computer before they are synthesized in experiments. We have predicted this material and now people are trying to synthesize the structure.” —Stefan Goedecker, University of Basel
Calculations suggest that the proposed structure, a hexagonal sheet, is chemically and mechanically stable and that its electronic structure can be modified by strain and defect engineering, giving it a tunable band gap that covers a wide energy range. It’s likely that the material can be deposited on various substrates without disrupting its structure.
The research—carried out by NCCR MARVEL’s Stefan Goedecker at the University of Basel and colleagues at the Institute for Advanced Studies in Basic Sciences and the University of Zabol in Iran and at Northwestern University in the U.S.—identified and characterized the novel structure thanks to an extensive search specifically aimed at screening 2D structures of titanium dioxide. The search was accelerated by machine learning methods that made it much less computationally intensive to carry out.
“What’s really important here is that we’ve used a scheme based on machine learning, which is some four orders of magnitude faster than density functional calculations,” Goedecker said. “The combination of that with structure prediction allowed us to work on a much larger scale than what was previously possible.”
The 2004 discovery of graphene and its extraordinary properties—notably its electronic structure, high mechanical strength, and excellent thermal conductivity—prompted an enormous amount of interest in 2D materials. Titanium dioxide, already used in industrial products ranging from consumer goods to advanced materials for photovoltaic cells, is one of the most versatile materials and can be produced in various nanostructures. Due to its promising photocatalytic, electrochemical and dielectric properties, a lot of research has been focused on finding ways to improve its performance by controlling size and/or shape of TiO2 at the nanoscale.
“I think it’s one of the most widely studied materials that exists,” Goedecker said. “What is surprising is that even for this widely studied material, we have found completely new structures that were previously overlooked.”
While people may have previously relied on chemical intuition to predict what sort of structures certain materials will adopt, the use of systematic search algorithms has revealed completely new and unexpected structures, he said. The approach also marks a difference to the way new materials are found. Until recently, it worked the other way around: scientists would find certain structures experimentally and then try to find a theoretical explanation.
“We’re now in a new era where we can make predictions and virtually discover new materials on the computer before they are synthesized in experiments,” Goedecker said. “We have predicted this material and now people are trying to synthesize the structure.”
The scientists’ approach to examining the atomistic structures of titanium dioxide also differs in that they changed the search algorithm, the so-called minima hopping method, to focus specifically on 2D structures. A lot of previous research has rather started with 3D structures, then cutting out certain planes to arrive at a 2D version. While the approach is understandable—using traditional density functional methods to do structure prediction on all of these possible materials would be too expensive—it assumes that atomically thin layers of titanium dioxide adopt structures close to that of the bulk material. Experiments have shown though that many 2D materials have structural features that strongly differ from those of their 3D counterparts. The structure discovered by Goedecker and his colleagues cannot be obtained by just cutting the bulk crystalline material in a certain way.
The exceptional properties of the proposed material suggest that it may be useful in energy applications, especially for photocatalytic hydrogen production because it has a large surface area with hexagonal voids and a favorable band alignment with respect to the redox potentials of water splitting. The structure may also be useful as a building block for 2D heterostructures, “sandwiches” of two or more semiconductor materials, that may be used in energy storage and conversion including metal-ion batteries and materials for photovoltaics.
“At the current state of research, we cannot claim that these materials will be specifically used for these applications,” said Maximilian Amsler, a researcher at Northwestern University and one of the study’s authors. “It’s rather hypothetical, but it could be useful.”
The next step, the scientists said, is actually synthesizing the material and Goedecker and Amsler are aware of two groups trying to do this. While there are some promising preliminary results, further investigations are required for conclusive experimental characterization.
Reference
Hossein Asnaashari Eivari, S. Alireza Ghasemi, Hossein Tahmasbi, Samare Rostami, Somayeh Faraji, Robabe Rasoulkhani, Stefan Goedecker, and Maximilian Amsler, Two-Dimensional Hexagonal Sheet of TiO2, Chemistry of Materials 29, 8594–8603 (2017), doi: 10.1021/acs.chemmater.7b02031
Low-volume newsletters, targeted to the scientific and industrial communities.
Subscribe to our newsletter